 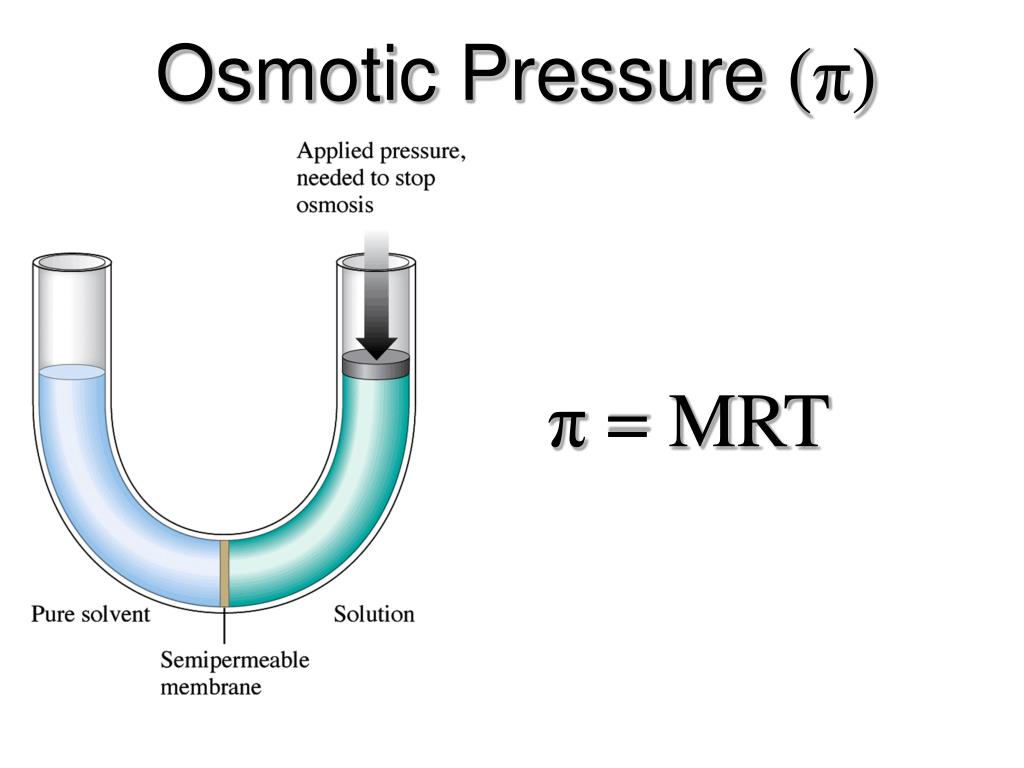
We get a mass here that is equal to 2.68 grams of hemoglobin. Five moles were given the molar mass here, which is 6.86 10 to the fourth grams per moon and this will yield 0.9 6 to 4. Let's sell for the mass of hemoglobin uh, which is here with a 3.90 times 10 to the negative. And then the volume is 100 milliliters of 1000.1000 Leaders, this will give us three points 90 times 10 to the negative five moles of hemoglobin. Hemoglobin, which would be 3.90 times 10 to the negative form moles per leader. Here we find that the polarity is 3.90 times 10 to the negative for Mueller. Kelvin temperature is 25 degrees Celsius, which is to 98 Calvin lips, all for the polarity. Osmotic pressure is expressed by the formula: iMRT (note how it resembles the PV nRT form of the Ideal Gas Law) where is the osmotic pressure in atm i van 't Hoff factor of the solute M molar concentration in mol/L R universal gas constant 0. It's convert this toe atmospheres so multiplied by 7 60 uh, this would be pointing Ain't 00 954 atmospheres 00954 atmospheres 0.8 to 06 Leaders atmospheres per mole. Osmotic pressure is 7.12 butts, 7.25 millimeters of Rick Green. First, let's use the osmotic formula for osmotic pressure rearranged itself from hilarity. Gnaiger E et al ― MitoEAGLE Task Group (2020) Mitochondrial physiology.Itself for the massive human woman. IUPAC Green Book 3rd Edition, 2nd Printing, IUPAC & RSC Publishing, Cambridge. ISBN 978-9-0Ĭohen ER, Cvitas T, Frey JG, Holmström B, Kuchitsu K, Marquardt R, Mills I, Pavese F, Quack M, Stohner J, Strauss HL, Takami M, Thor HL (2008) Quantities, Units and Symbols in Physical Chemistry. For historical reasons and for non-molecular compounds, such as ionic salts, it is still frequently used.īureau International des Poids et Mesures 2019 The International System of Units (SI)īureau International des Poids et Mesures (2019) The International System of Units (SI). The formula weight is another synonym of the molar mass that is not covered by the IUPAC definition. 
Despite this definition, the term "molecular weight" is widely used as a synonym for the molar mass. Round your answer to 3 significant digits mol Check. The osmotic pressure of this solution is measured to be 0254 atm at 25.0 C Calculate the molar mass of the protein. mg of an unknown protein are dissolved in enough solvent to make 5.00 mL of solution. The relative molar mass and the molecular weight are alternative terms for the relative molecular mass according to IUPAC's Green Book or Gold Book. Using osmotic pressure to find molar mass 381. Since mass is divided by mass, the relative molecular mass is dimensionless. It is the mass of an entity (this can be a molecule or a formula unit: a group of atoms by the way the chemical formula is written) divided by the unified atomic mass unit. The relative molecular mass is represented by the symbol M r and defined as M R = m f/ m u. However, for historical as well as usability reasons, g It is calculated as the sum of standard atomic weights of all atoms that form one entity of the substance. ScienceChemistryQ&A LibraryWhat is the molar mass of a protein if a solution of 0.02 g of the protein in 25.0 mL of solution has an osmotic pressure of 0.56. The molar mass allows for converting between the mass of a substance and its amount for bulk quantities. The definition applies to pure substance. So, you can begin with the standard equation for osmotic pressure: osmotic pressure () iMRT 0.125 atm i van't Hoff factor 1 for a polypeptide since it does not ionize or dissociate M molarity moles of polypeptide / liter of solution R gas constant 0. At 23☌ the solution has an osmotic pressure of 0.118 mm Hg. A 20.0-mg sample of this enzyme is dissolved in enough water to make 225 mL of solution. Lysozyme, extracted from egg whites, is an enzyme that cleaves bacterial cell walls. It is defined as M B = m/ n B, where m is the total mass of a sample of pure substance and n B is the amount of substance B given in moles. EXAMPLE 14-9 Establishing a Molar Mass from a Measurement of Osmotic Pressure. Molar mass M is the mass of a chemical compound divided by its amount-of-substance measured in moles. High-resolution terminology - matching measurements at high-resolution
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
